Microbiological Culture Media: Halal risk in FMCG production facility
Halal food production is about more than simply avoiding certain food sources during production. The authorities that certify and recognize halal food products require adherence to a wide range of other guidelines. This article discusses and highlights the halal culture media usage & concept as one of the halal-risk measurements in food & beverage (F&B), cosmetics and pharma manufacturing quality assurance.
Ensuring raw material and ingredient sources are halal and safe is one of the requirements listed under the Malaysia Halal Certification Procedure published by JAKIM. There are many different types of raw materials that production plant & microbiology or QA/QC laboratory use to create & test finished products, including culture media for microbiological testing.
Watch on-demand webinar-Halal Industry Awareness
Raw materials & ingredients of culture media
Culture medium or growth medium is a liquid or gel designed to support the growth of microorganisms. A typical culture media contains bio-sourced ingredients such as extracts, enzymes, sugar and protein-based materials. These components are originated from different type of meat sources including porcine, bovine, poultry or plants. On top of that, enzymes used for protein (tryptone, peptone, etc.) hydrolysis in the culture media content can be derived from animal sources such as porcine or bovine.
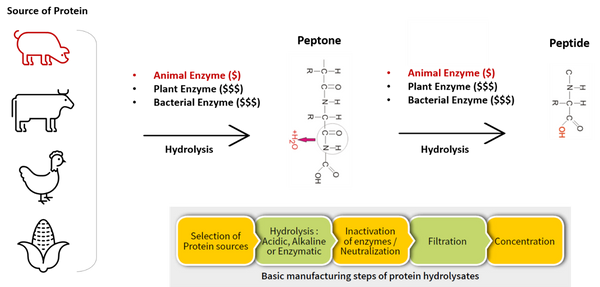
Both the Halal Committee and QA/QC team should look beyond than just the quality and price of culture media. Give more attention when inspecting and questioning your culture media supplier, especially on the raw materials, source of origin and related certifications.
What to look for when conducting Halal checklist at your facility?
- List of raw materials used in both laboratory and manufacturing site
- “Spot-check” of common and non-common culture media
- Culture media manufacturer - plant Halal certificate
- Halal Certification & Accreditation bodies that recognized by JAKIM
Culture media in contact with production line
Microbiological culture media such as harmonized tryptone soya broth and linden grain medium is commonly used for media fill run test for pharma aseptic production process and CIP setup of beverage production, respectively. Good Manufacturing Practices (GMP) require pharmaceutical companies to regularly perform media fill tests in order to verify the adequate microbiological state of their aseptic production process. Media fill tests are critical microbiological tests carried out to simulate the normal manufacturing conditions by replacing the pharmaceutical product with culture media.
Commonly, sponges or swabs are used to collect samples from both food contact surfaces (e.g., slicers, mixers, utensils or conveyors) and non-food contact surfaces (e.g., floors, drains, carts or equipment housing) for routine environmental monitoring program practices. However, when sampling is conducted on surfaces previously exposed to chemical germicide treatment, appropriate neutralizers must be incorporated into the sponges or swabs to preserve viability of the microbial cells. Neutralizers or culture broth recommended for food plant monitoring include Dey-Engley neutralizing broth (DE), neutralizing buffer (NE), Buffered peptone water (BPW) and Letheen broth (LT). For pharmaceutical industry, contact plates are the standard devices for environmental monitoring of surfaces and personnel in cleanrooms and isolators. The most commonly used media for this purpose is tryptic soy agar (TSA), a non-selective medium known for its ability to allow a broad variety of organisms to grow.

At FC-BIOS, we believe that every point matters, including the usage of Halal microbiological culture media to meet the guidelines of Malaysia Halal standard. HiMedia products are inspected and assessed by Jamiat Ulama-i-Hind Halal Trust, recognized by the Department of Islamic Development Malaysia (JAKIM), to ensure Halal compliance and finished product integrity.
Download newsletter: Himedia Halal Certified Microbiological Media